Flow Cytometric Analysis of Cerebrospinal Fluid (CSF)
Invasion of leucocytes into cerebrospinal fluid has grave prognostic significance and requires important therapeutic decisions including the administration of intrathecal chemotherapy. Leptomeningeal localisation is conventionally diagnosed by cytomorphological analysis through identification of malignant lymphocytes in CSF, however this technique has a relatively high rate of false-negative results in up to 60% of cases. Recent reports suggest that multiparameter flow cytometric assessment of CSF samples could improve the efficiency of detection of CNS involvement, due to its high specificity and greater sensitivity, however low cell counts and rapid decline of cells after lumbar puncture can pose technical challenges for flow cytometric analysis (1).
TransFix has been used for the stabilisation of malignant haematological cells found in CSF. TransFix/EDTA CSF Sample Storage Tubes contain TransFix that has been optimised for CSF stabilisation and have been recommended in guidelines for CSF sample storage, published in the British Journal of Haematology (see case study 1).
TransFix has also been shown to reduce cellular lysis after 30 minutes compared to fresh CSF (see case study 2) and to stabilise key CSF leucocyte markers for up to 72 hours (see case study 3). The stabilisation process preserves light scatter and key antigen expression patterns allowing analysis of diagnostic and follow up CSF specimens for patients with CNS infiltration (see case study 3 and case study 4).
- Case Study 1: Guidelines on the use of multicolour flow cytometry in the diagnosis of haematological neoplasms
- Case Study 2: Use of TransFix Cerebrospinal Fluid (CSF) Storage Tubes Prevents Cellular Loss and Enhances Flow Cytometric Detection of Malignant Hematological Cells After 18 Hours of Storage
- Case Study 3: Infiltration of CNS by acute leukaemia: Analysis of fresh and stabilised CSF
- Case Study 4: CSF Screening for Suspected CNS localised leukemia or lymphoma
Features and benefits of CSF stabilisation for Flow Cytometry
- Potential to reduce the number of repeat lumbar punctures required due to degraded samples.
- TransFix/EDTA CSF Sample Storage Tubes are specifically designed for the stabilisation of cells within CSF specimens.
- Quality controlled against CSF substitute, including the markers CD14, CD4, CD8, CD19, CD20, Igκ, Igλ, CD56. (see CoAs for full details).
- Specific protocol for CSF sample preparation with TransFix (see IFU).
- Higher lymphocyte yield after 30 minutes compared to untreated samples (see case study 2).
- The cellular component of CSF is stabilised for at least 72 hours allowing for determination of cell sub-populations and follow up analysis (see case study 3).
- Light scatter and key antigen expression patterns are comparable to fresh samples (see case study 3and case study 4).
- Markers stabilised include Markers stabilised include CD10, CD19, CD34 (see case study 3 and case study 4), CD45, HLA-DR, and CD117 (see case study 3).
Case Study 1: Guidelines on the use of multicolour flow cytometry in the diagnosis of haematological neoplasms
These guidelines have been developed by UK-based clinical flow cytometry (FC) experts along with clinical representatives to take into account advances in the state of the art of clinical flow cytometry since 2002. Their work was revised by the Haemato-oncology Task Force of the British Committee for Standards in Haematology (BCSH) before review by other UK-based Haematologists and FC experts, the BCSH and the British Society for Haematology committee. This document covers a varied range of topics, from selection and setup of flow cytometers through data analysis to training considerations for staff working in the clinical FC laboratory.
The guidelines strongly recommend sample age and quality is assessed prior to analysis and in the case of cerebrospinal fluid (CSF), it is recommended that unless immediate analysis is possible then samples can be stored in TransFix/EDTA CSF Sample Storage Tubes for up to 72 hours before analysis
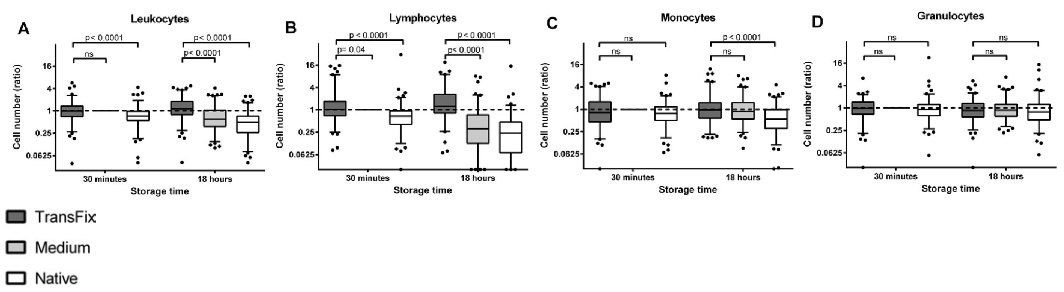
Case Study 2: Use of TransFix Cerebrospinal Fluid (CSF) Storage Tubes Prevents Cellular Loss and Enhances Flow Cytometric Detection of Malignant Hematological Cells After 18 Hours of Storage
In patients with suspected or proven leptomeningeal localised haematological malignancies (LHM) in CSF, rapid assessment is required as cell numbers decline quickly after lumbar puncture. The study aim was to examine leucocyte numbers and detection of LHM by flow cytometry in 99 CSF samples from patients with suspected or proven LHM after 30 minutes and 18 hours of storage in i) TransFix/EDTA CSF Sample Storage Tubes ii) serum-containing medium and iii) no stabilising agent (untreated). It was found that leucocyte numbers in TransFix stabilised CSF were higher than in the corresponding untreated samples at both time points. After 18 hours of storage at 4°C, the detection of LHM was enhanced in TransFix stabilised CSF. For all discordant paired observations, the level of suspicion of a patient having LHM was higher for the TransFix stabilised samples than for the untreated samples. The authors concluded that the use of TransFix/EDTA CSF Sample Storage Tubes prevents cellular loss and enhances flow cytometric detection of LHM after 18 hours of storage.
Case Study 3: Infiltration of CNS by acute leukaemia: Analysis of fresh and stabilised CSF
A research study to determine whether acute leukaemia blasts present in CSF can be successfully stabilised by TransFix has concluded that Transfix preserved light scatter and key antigen expression patterns allowing analysis of diagnostic and follow up CSF specimens for patients with CNS infiltration.
Full evaluation of the antibody panels to be used on transfixed samples is also required since some antigens are expressed dimly.
Transfix-induced light scatter changes do not impair cell identification
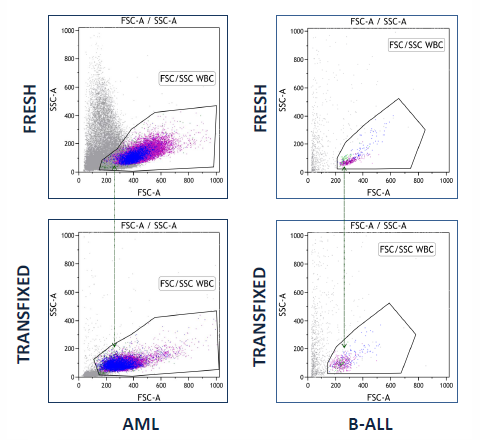
TransFix maintains key B-ALL antigen expression
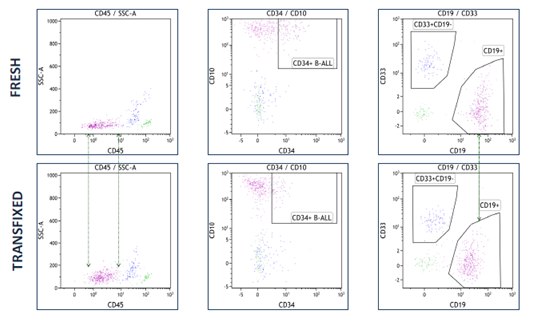
TransFix maintains key myeloid antigen expression
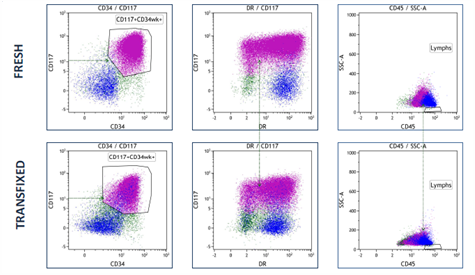
In collaboration with Haematology Dept, Bristol Royal Infirmary, University Hospitals Bristol NHS Foundation Trust, UK.
Case Study 4: CSF Screening for Suspected CNS localised leukemia or lymphoma
Evaluation studies into the use of TransFix/EDTA for stabilising CSF samples from patients with suspected CNS localised leukemia or lymphoma are being conducted*. Results to date indicate the following:
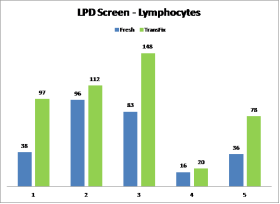
- Lymphocyte cell recovery of most subsets observed in TransFix/EDTA treated CSF samples matched or exceeded that of the fresh samples (Graph 1).
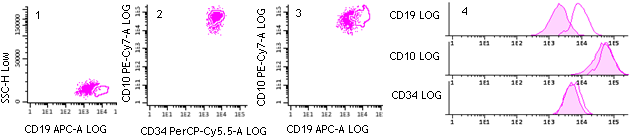
- Antigen expression profiles in TransFix/EDTA treated CSF are maintained for 3-7 days and are comparable to equivalent fresh, untreated CSF samples (Figure 1-4).
- The diagnostic conclusion would have remained the same for all patients using TransFix/EDTA treated samples compared to fresh (Figures 1-4).
- Results suggested that TransFix/EDTA can be used successfully to stabilise CSF samples for screening purposes.
Lymphocyte cell recovery within TransFix treated & untreated CSF samples
B-blast cells in a TransFix treated & untreated CSF sample
TransFix Product Format
 TransFix/EDTA CSF Sample Storage Tubes contain a TransFix and EDTA mix that has been optimised for stabilizing key CSF leucocyte markers for up to 72 hours.
TransFix/EDTA CSF Sample Storage Tubes contain a TransFix and EDTA mix that has been optimised for stabilizing key CSF leucocyte markers for up to 72 hours.
For more information on CSF Stabilisation, please contact us!